Improving Pharmaceutical Operations
Your all-in-one EHSQ Connected Worker platform enables you ensure high-quality and safety-compliant operations while supporting frontline workers with digital guidance.
Schedule Demo



Transform paper procedures with a single EHSQ Connected Worker solution to increase safety, guarantee real-time visibility, support frontline workers' decision-making and improve operational results.
Embed validated procedures into cleanroom and lab operations.
Ensure secure, traceable, and compliant documentation aligned with FDA and EMA expectations.
Structure investigation workflows to prevent repeat compliance failures.
Digitally capture environmental parameters and alert deviations.
Standardize validation workflows for regulated production assets.
Learn how a leading global healthcare company specialized in pharmaceutical products leveraged the EHSQ Connected Worker solution to improve execution compliance by 96%.

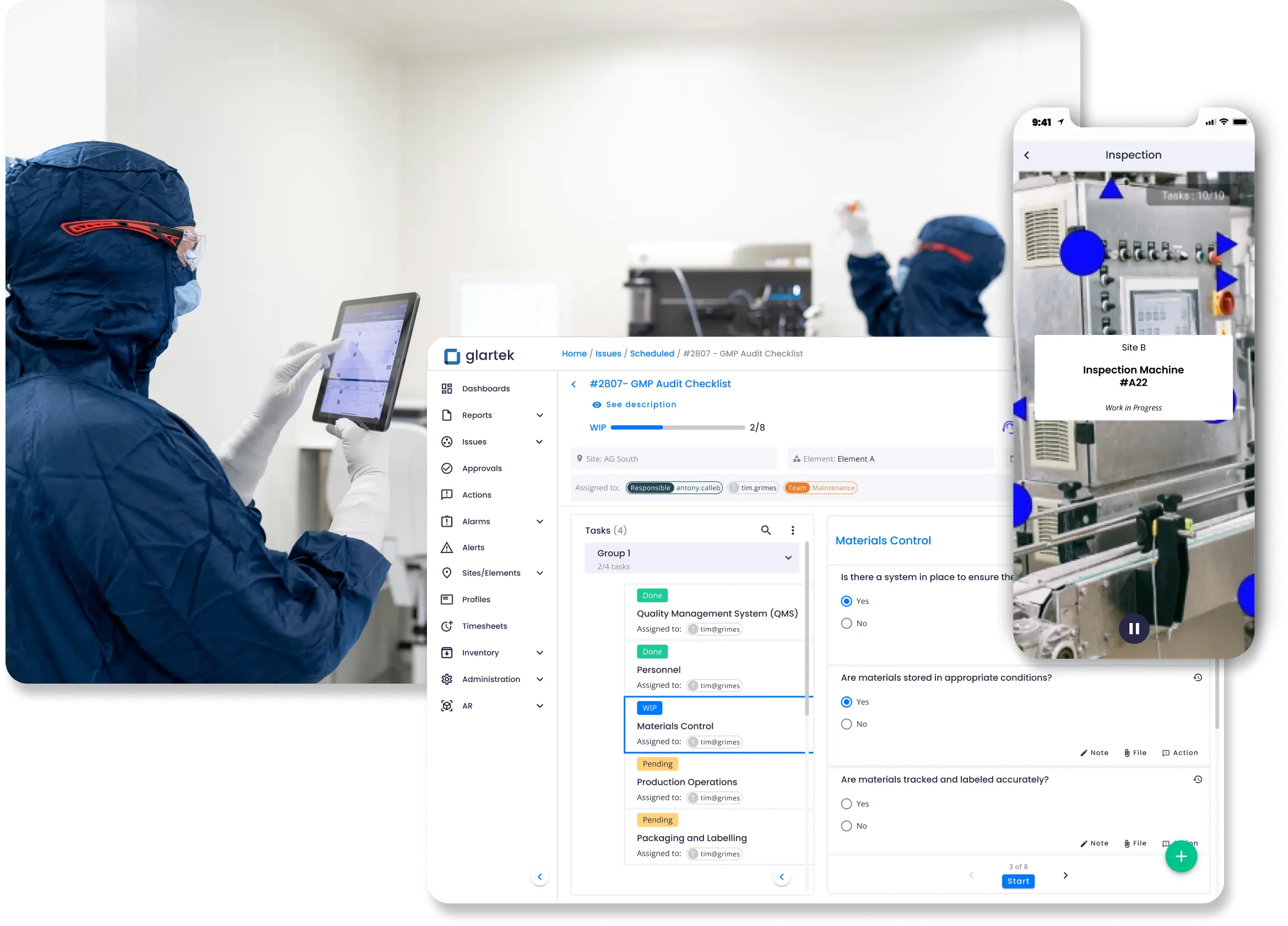
Comply with strict regulations by monitoring operations against GxP requirements to detect non-conformities early. Fully tracked processes create clearer, more efficient audit trails.
- Improved regulatory confidence
- Reduced compliance risk
- Faster, more reliable audits
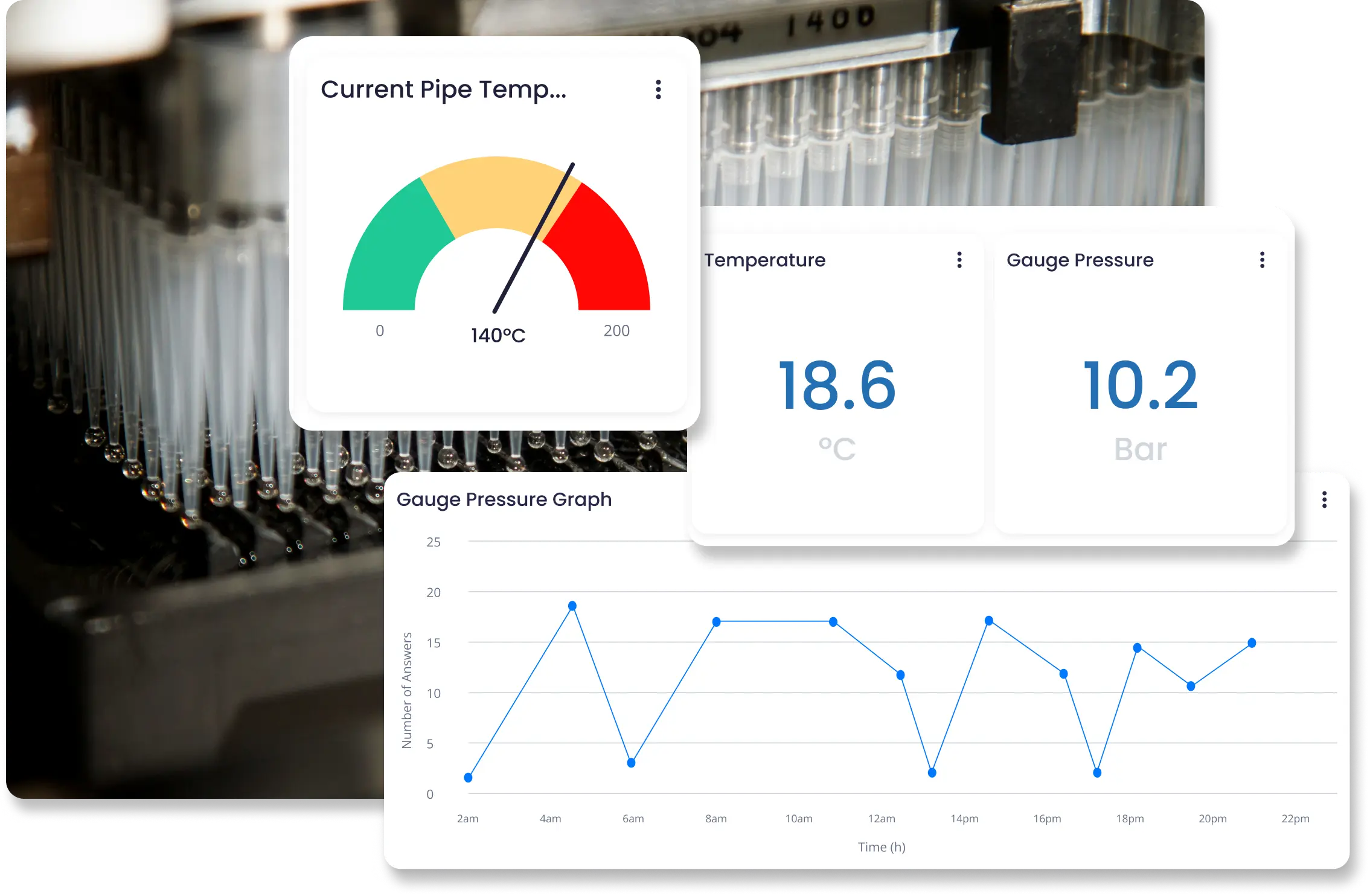
Continuously monitor procedures, equipment, and laboratory conditions using predictive analytics and instant alerts to catch risks early. Respond to potential hazards before they escalate.
- Early detection of operational issues
- Faster hazard response and resolution
- Stronger laboratory safety control
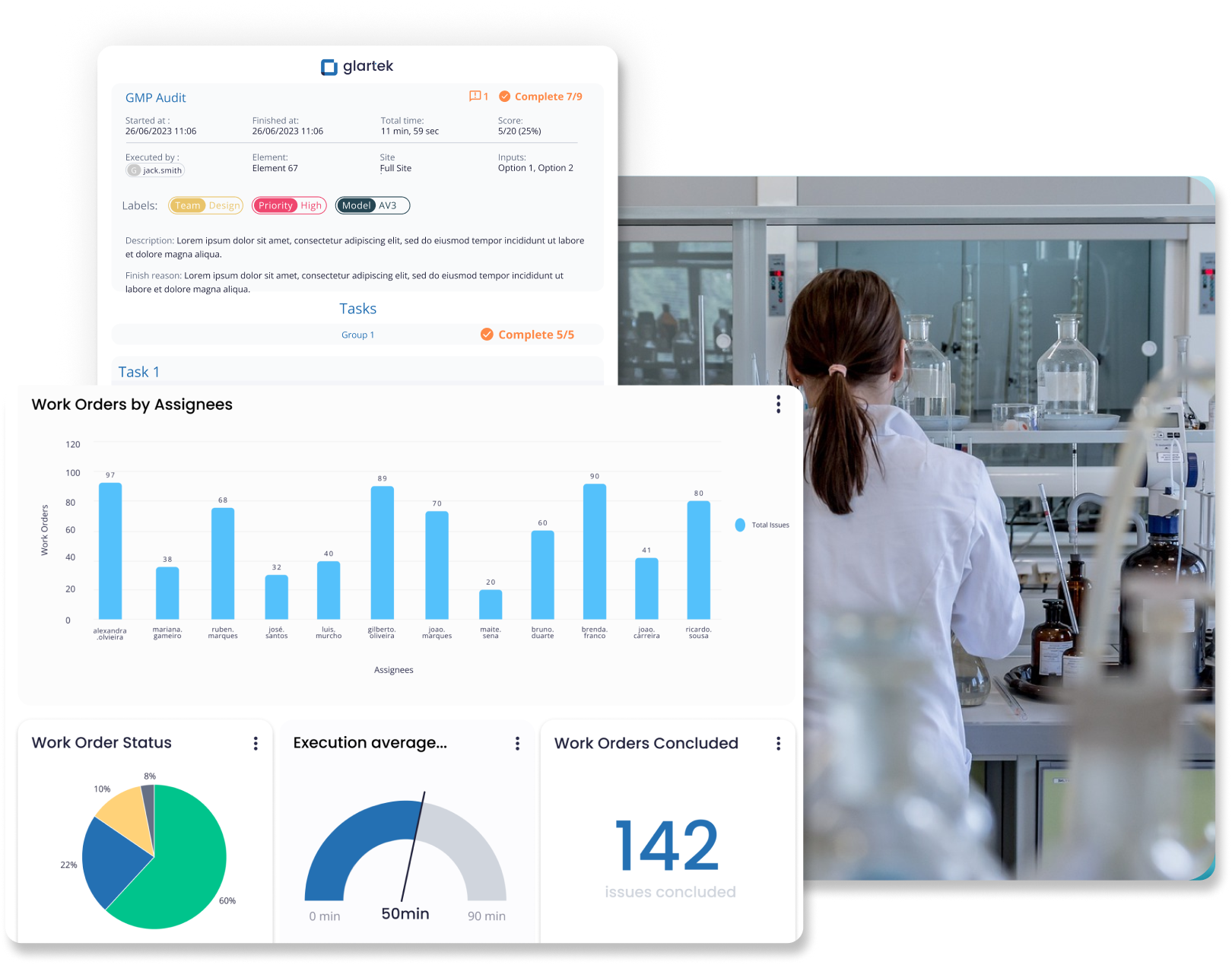
Maintain compliance with strict regulations by continuously evaluating GxP requirements to identify non-conformities early. Fully tracked and recorded processes make audit trails clearer and more efficient.
- Improved regulatory assurance
- Reduced compliance risk
- Faster, easier audits
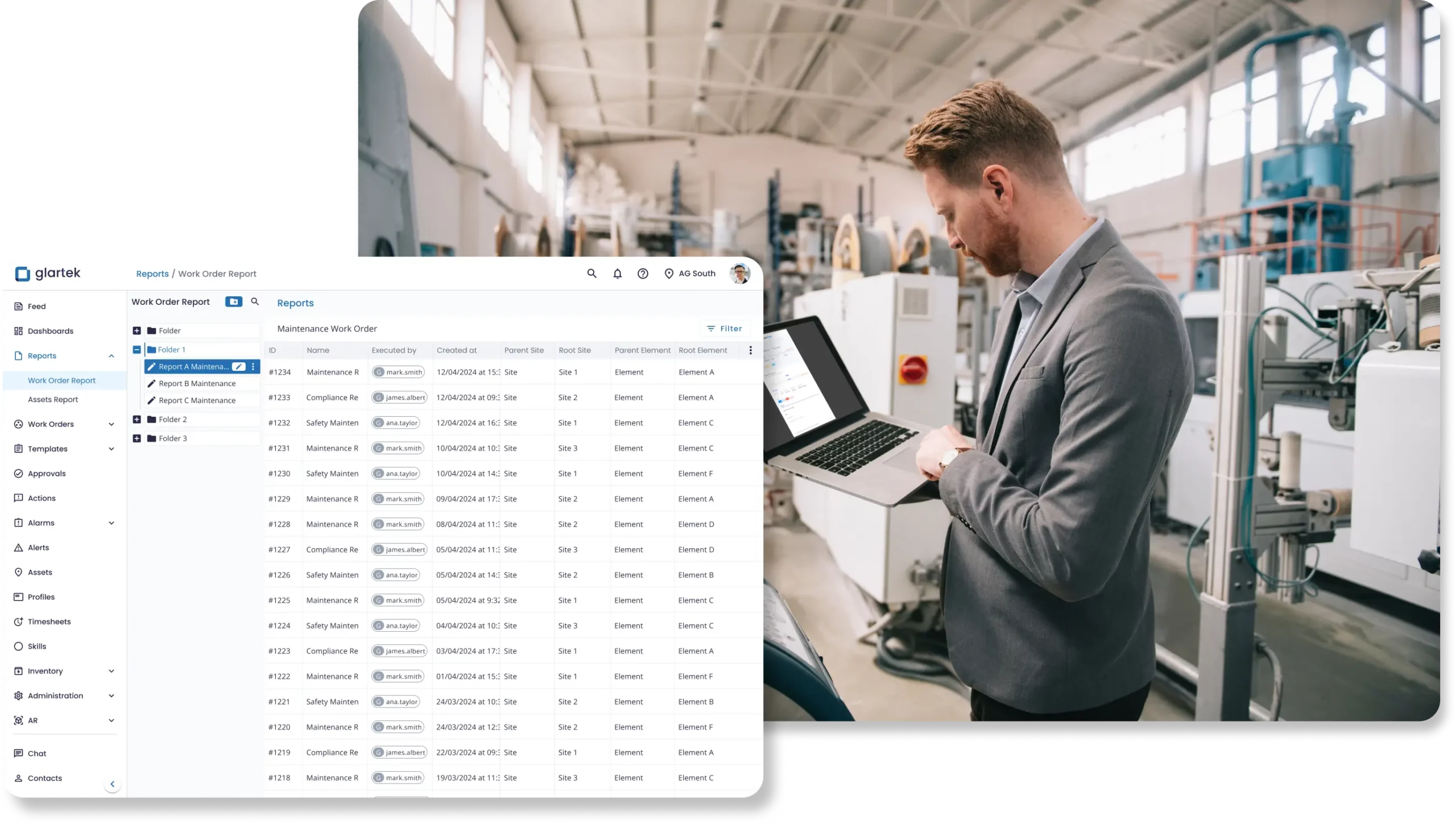
Access real-time data and status updates to speed approvals and streamline processes. Use dashboards and reports to make faster, smarter, data-driven decisions.
- Faster approvals and workflows
- Improved operational efficiency
- Data-driven decision-making

“With Glartek's platform, we are finally able to keep our most experienced employees centralized, and consequently, our experts are able to solve more customers’ requests than before. Fortunately, both our junior workers and customers are learning how to perform operations they didn’t know before, which improved our operational efficiency and results.”
Dr. Radek Řezáč, Operations Director
All in one platform
Ensure pharmaceutical compliance requirements like GMP, GDP, and more with pre-configured templates.
Easily configure and adapt workflows to meet evolving regulatory requirements with a no-code template builder.
Gain instant visibility into processes, equipment status, and quality metrics with clear dashboards and reports.
Protect sensitive information with security access controls (SSO, role-based permissions) and be compliant with data privacy regulations.
Request your demo
Start your EHSQ Connected Worker journey with Glartek and become a leader in your industry.
Schedule Demo.webp)
Discover the power of the only AI-Native EHSQ solution built for the frontline