Operações farmacêuticas otimizadas
Plataforma completa de EHSQ (SSMA) e Connected Worker: Garanta a conformidade da sua operação com suporte digital e orientação em tempo real para as equipes de campo
Agende uma demonstração



Elimine papéis e processos manuais definitivamente. A plataforma da Glartek permite que o colaborador de campo realize cada registro, inspeção e conformidade técnica de forma instantânea via dispositivo móvel. Garanta visibilidade em tempo real para a gestão e suporte digital para as equipes de operação na indústria farmacêutica.
Integre procedimentos validados às operações em salas limpas e laboratórios.
Garanta uma documentação segura, rastreável e em conformidade com as exigências da ANVISA, FDA e EMA.
Estruture os fluxos de trabalho de investigação para evitar não conformidades recorrentes.
Registre digitalmente parâmetros ambientais e desvios de limites de alerta.
Padronize os fluxos de validação para equipamentos e sistemas sujeitos à regulamentação.
Descubra como uma líder global em saúde otimizou seus processos com nossa solução de SSMA e Connected Worker, alcançando 96% de conformidade na execução.

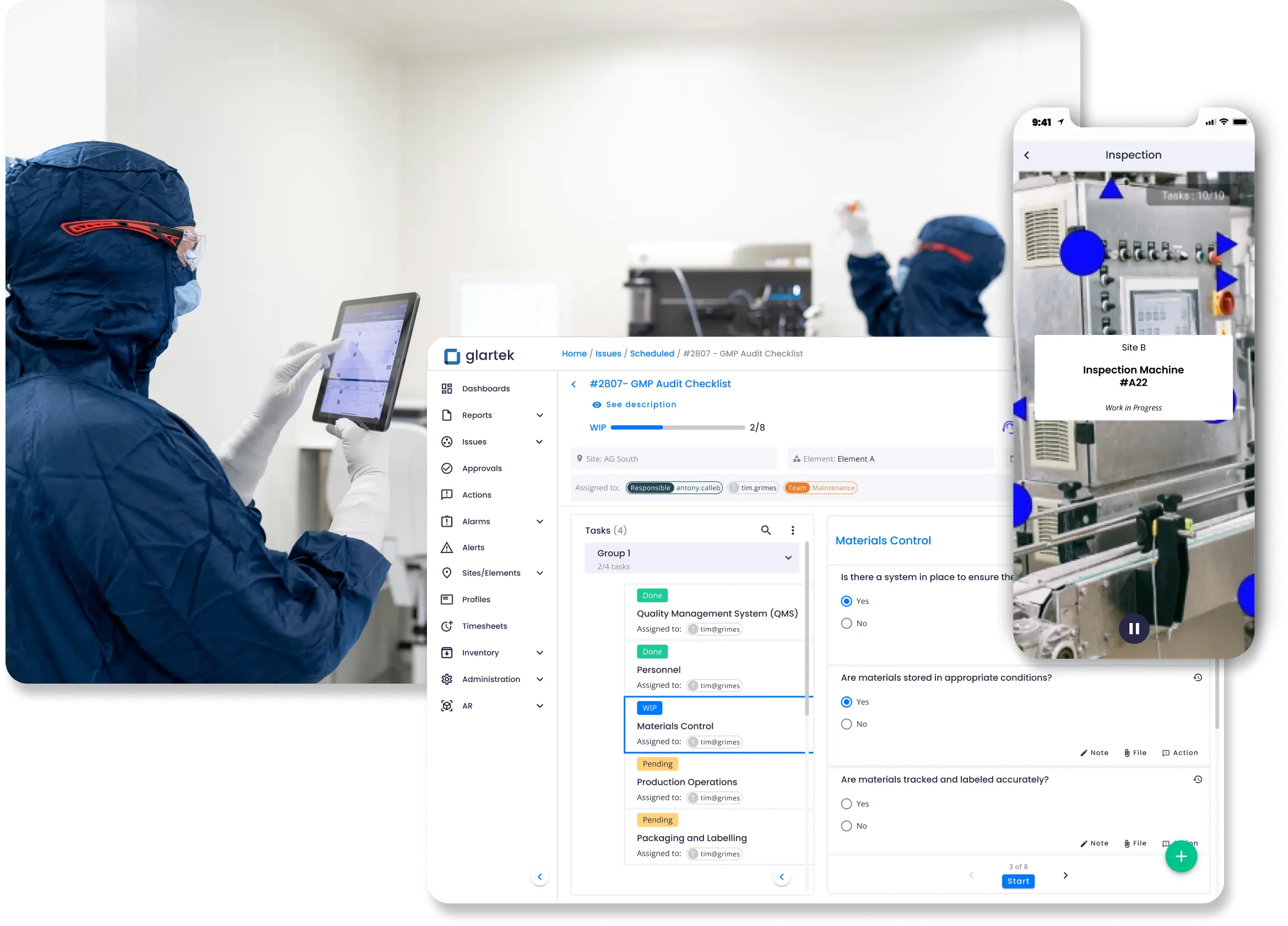
Atenda a regulamentações rigorosas monitorando as operações em tempo real conforme os requisitos de GxP. Identifique antecipadamente as não conformidades e crie trilhas de auditoria automatizadas, claras e seguras.
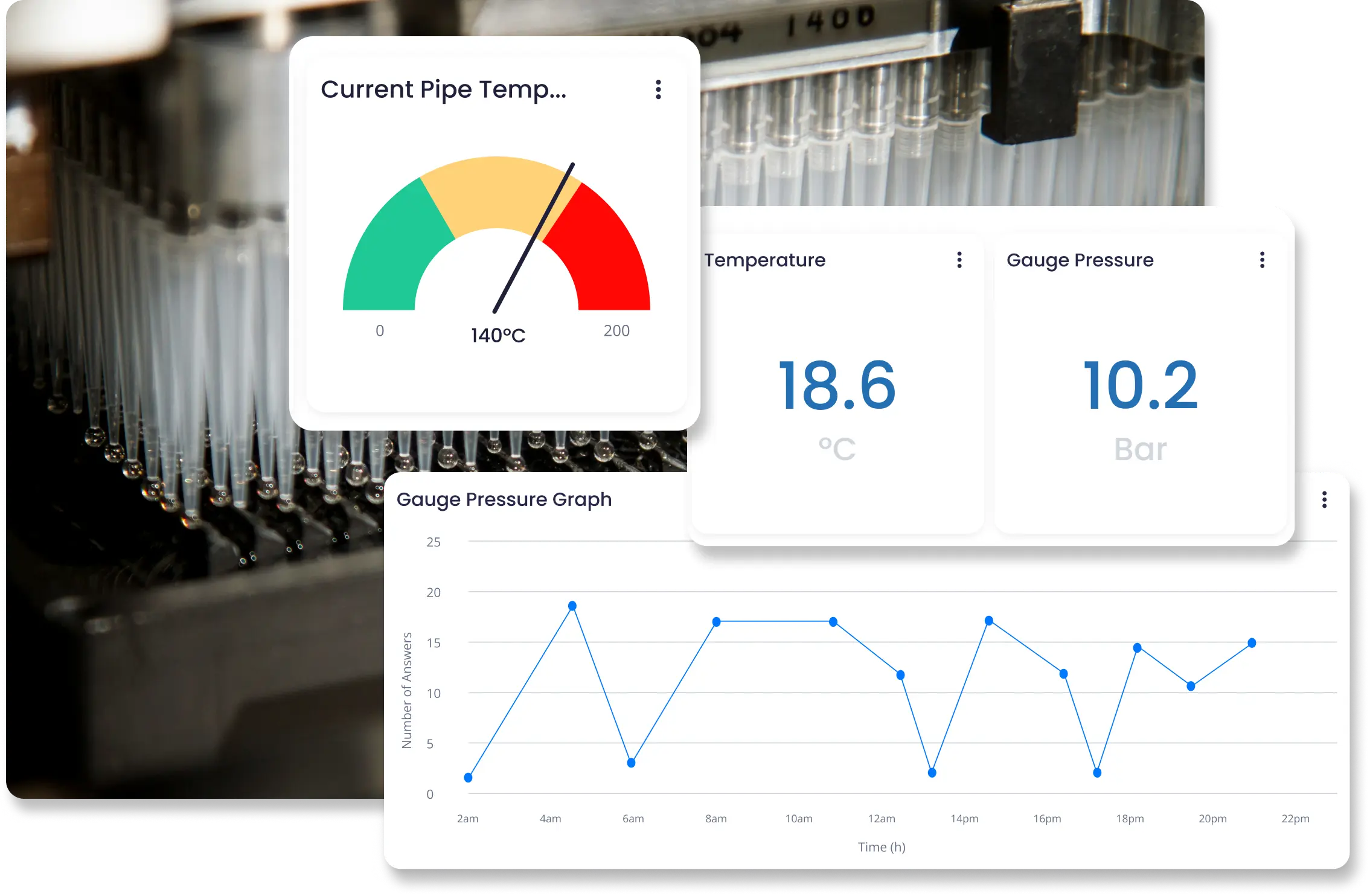
Mantenha a conformidade com regulamentações rígidas avaliando continuamente os requisitos de GxP para identificar antecipadamente não conformidades. Processos totalmente monitorados e registrados tornam as trilhas de auditoria mais claras e eficientes.
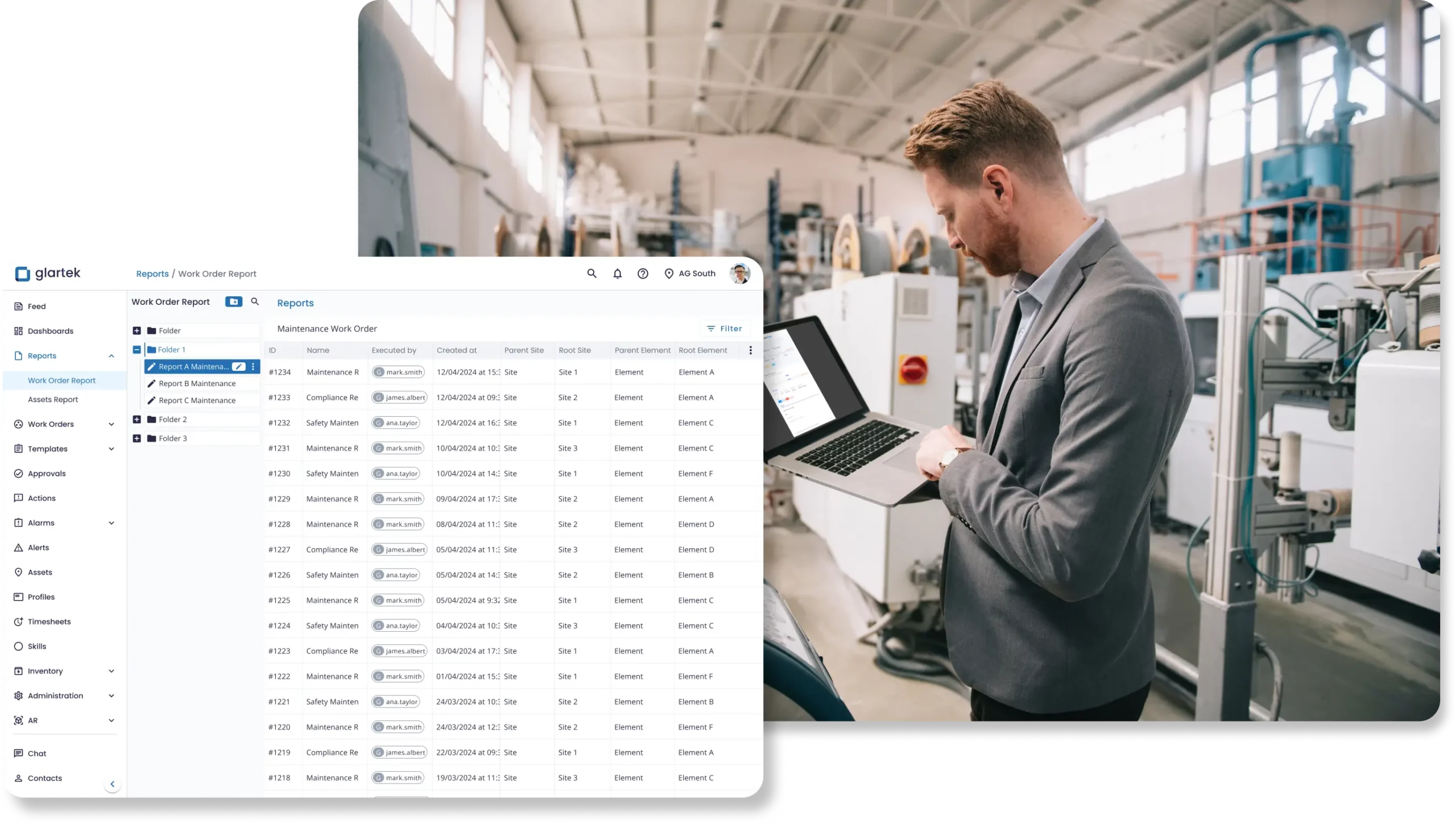
Acesse dados em tempo real e atualizações de status para acelerar aprovações e otimizar processos. Utilize dashboards e relatórios para tomar decisões mais rápidas, inteligentes e totalmente baseadas em dados.
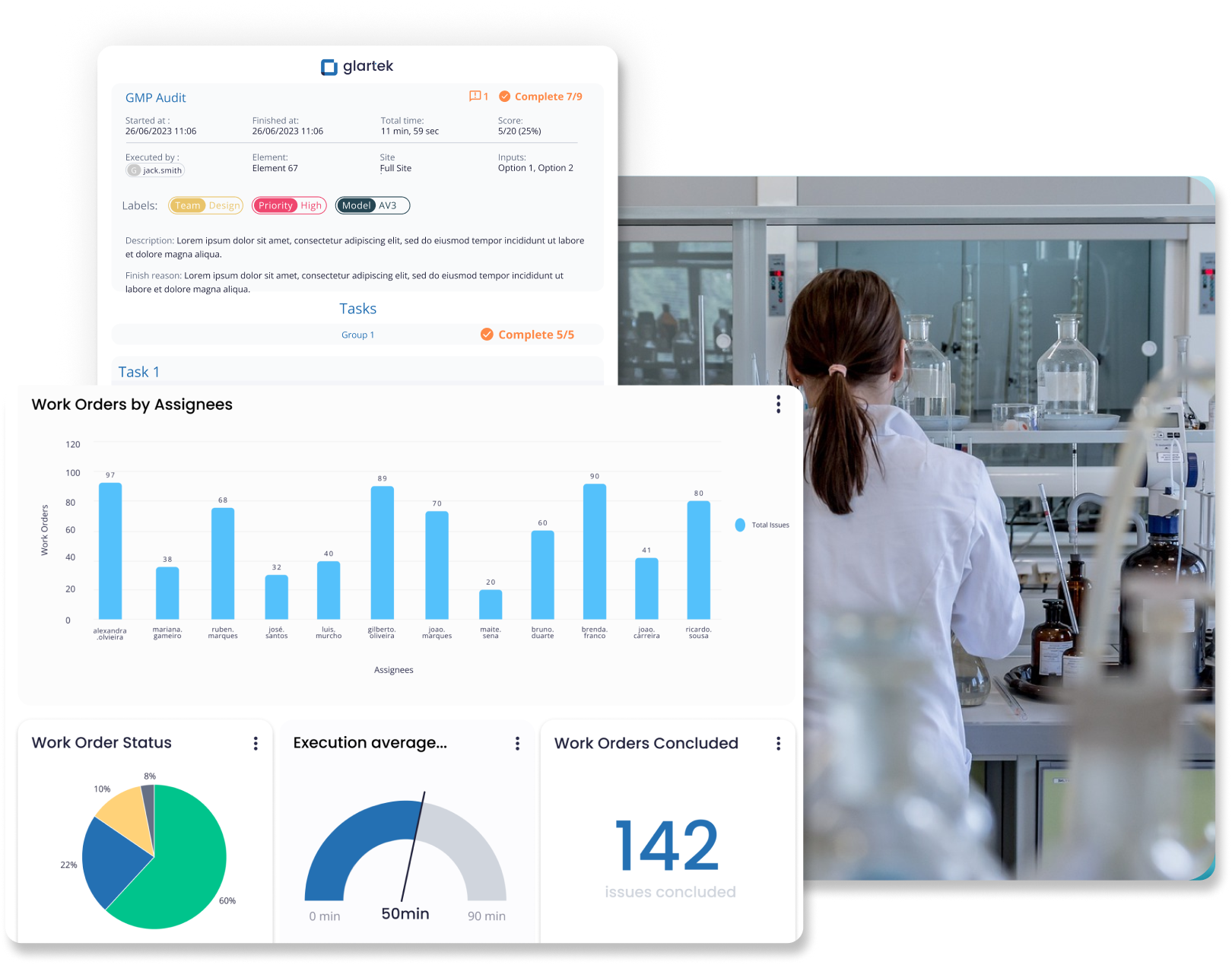

“Com a plataforma da Glartek, conseguimos centralizar o conhecimento dos nossos especialistas e, com isso, eles agora resolvem um volume de chamados muito maior. Além disso, nossos técnicos juniores e até nossos clientes estão capacitados para realizar operações complexas que antes desconheciam, o que elevou nossa eficiência operacional e nossos resultados financeiros.”
Dr. Radek Řezáč, Diretor de Operações
Tudo em uma única plataforma
Garanta o cumprimento dos requisitos regulatórios farmacêuticos (como GMP/BPF, GDP/BPD, entre outros) utilizando modelos pré-configurados.
Configure e adapte fluxos de trabalho com agilidade para atender à evolução das normas regulatórias, utilizando um editor de modelos no-code.
Obtenha visibilidade instantânea dos processos, status dos equipamentos e métricas de qualidade por meio de dashboards e relatórios detalhados.
Proteja informações sensíveis com controles de acesso rigorosos (SSO, permissões baseadas em funções/perfis) e garanta total conformidade com a LGPD e outras regulamentações de privacidade.
Solicite uma demonstração
Inicie sua jornada em EHSQ (SSMA) com a Glartek e torne-se um líder no seu setor.
Agende uma demonstração.webp)
Descubra o poder da única Solução EHSQ (SSMA) nativa em IA desenvolvida para a linha de frente