Gestão da qualidade
Gerencie auditorias, inspeções, observações, CAPAs e controle de mudanças em uma única plataforma. A solução da Glartek foi criada para apoiar as equipes no atendimento aos requisitos da ISO e na redução de reincidências em todas as unidades.
Solicite uma demonstração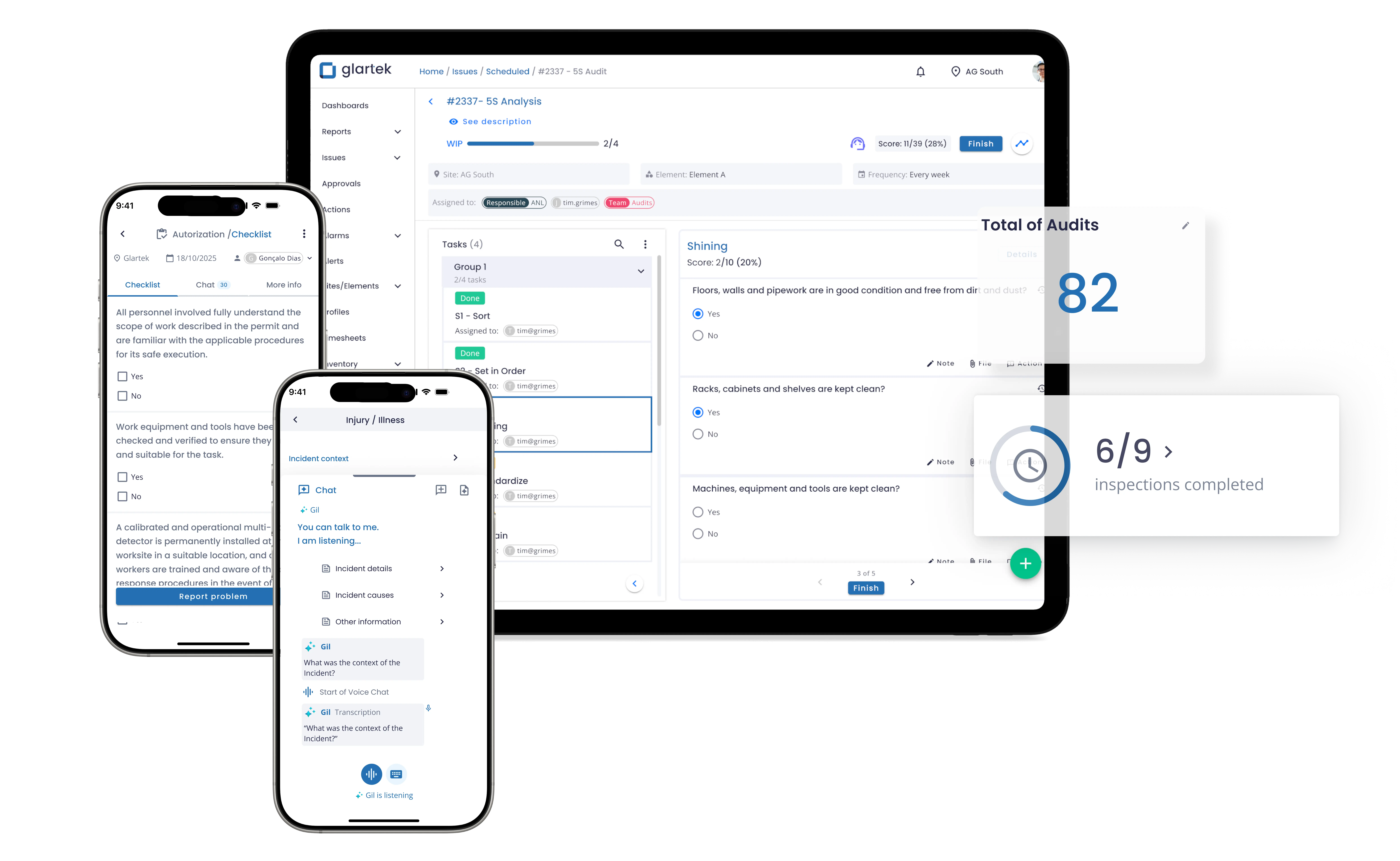
Desafio
Espera-se que as equipes de qualidade antecipem problemas antes que ocorram. No entanto, o desafio é gerenciar processos complexos, unidades distribuídas geograficamente e evidências dispersas em diversos sistemas.
Auditorias, inspeções e ações corretivas costumam ser geridas em ferramentas isoladas, dificultando o vínculo entre as constatações e a resolução definitiva.
O acompanhamento de objetivos, responsabilidades e progressos normativos torna-se complexo quando evidências e atualizações não estão centralizadas.
As investigações costumam ser demoradas e variam entre unidades, resultando em diagnósticos inconsistentes e ações que não eliminam a causa real dos desvios.
Frequentemente, as ações corretivas (CAPAs) permanecem abertas além do prazo ou são encerradas sem a devida verificação de eficácia, elevando o risco em auditorias e permitindo a reincidência de desvios.
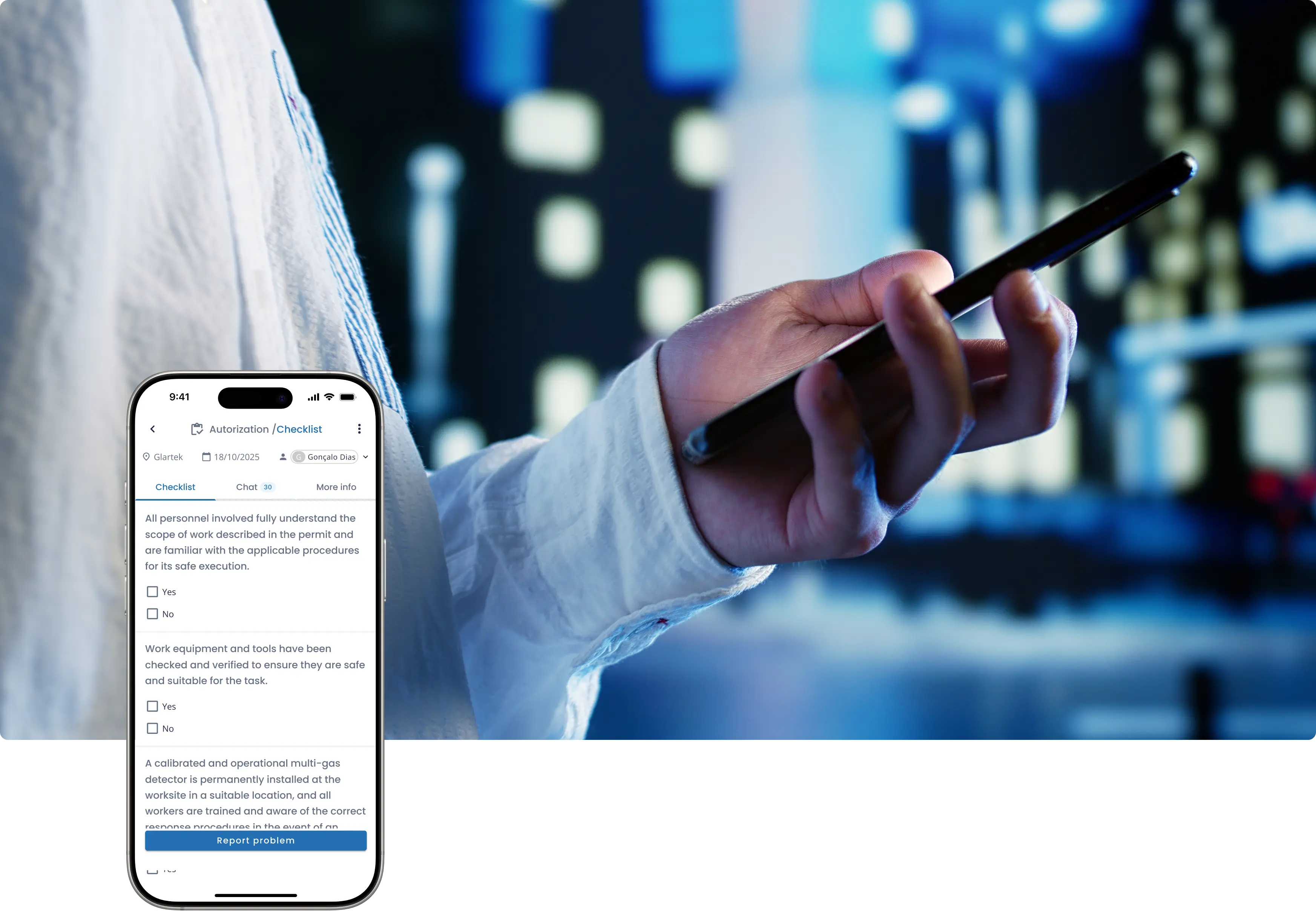
Gerenciar alterações via e-mail e planilhas causa atrasos, perda de aprovações e rastreabilidade limitada — justamente nos processos onde o controle de risco e a conformidade são mais críticos.
A única solução EHSQ criada para a linha de frente com impacto direto no gerenciamento

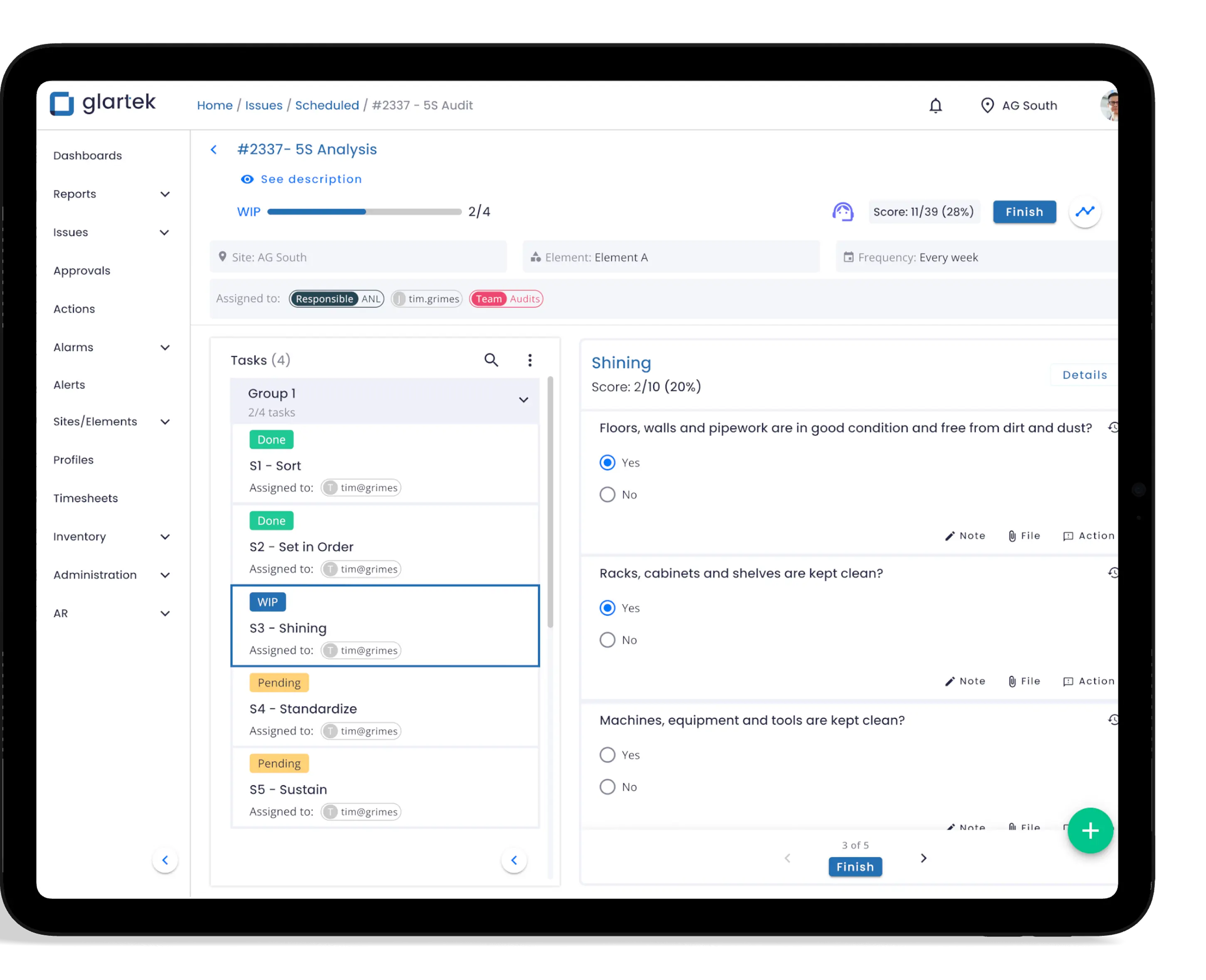
Prontidão para Auditorias (Audit-Ready)

Agilidade em Investigações e Soluções

Conformidade robusta e melhoria contínua
Explorando todas as nossas soluções
Mantendo a qualidade no dia a dia
45%
Mais rapidez no fechamento de CAPAs
52%
Redução de tempo na preparação para auditorias
28%
Menos reincidências de não conformidades
33%
Redução em incidentes registráveis




%201.webp)

Depoimentos
“A solução da Glartek nos permite fazer um diagnóstico mais fácil e rápido dos problemas que temos em nossas operações. A solução foi muito eficaz e atendeu às nossas expectativas; portanto, no futuro, nosso objetivo é expandi-la para nossas diferentes atividades.”
%201.webp)
Qualidade operacional
Quando os fluxos de gestão da qualidade são padronizados, a melhoria contínua deixa de ser teórica e se torna mensurável. Organizações que digitalizam auditorias, inspeções, RCA e CAPA eliminam gargalos operacionais, garantem a rastreabilidade total e mitigam a reincidência de não conformidades, integrando cada etapa, desde o relato da primeira observação até a verificação final de eficácia.
Solicite uma demonstraçãoDescubra o poder da única Solução EHSQ (SSMA) nativa em IA desenvolvida para a linha de frente